Alopecia Areata (AA) is an autoimmune disease that causes hair loss in clumps. It is characterized by rapid progression of the hair follicle from anagen (growing) to telogen (resting) phase, and a collapse of immune-privilege that is normally enjoyed by the bulb of the hair follicle. The loss of immune-privilege opens up the hair follicle to attack by cytotoxic CD8+ immune T cells, leading to hair loss. Clinically, AA manifests as a distinct pattern, most commonly as small round/patchy bald lesions on the scalp that can progress to total loss of scalp hair. AA affects about 2% of the general population at some point during their lifetime, and it is the second most frequent form of alopecia after male- and female-pattern baldness.
SCD-153 has the potential to be the first topical therapy approved for AA, and as a topical agent is expected to have comparatively less side-effects than the approved oral therapies.
Mechanistic studies in vitro have shown that SCD-153 significantly attenuates expression of CXCL9, 10, and 11, and IL-1β, IL-6 and IFN-β, when exposed to human epidermal keratinocytes, human outer root sheath cells or live human skin that is stimulated with polyinosinic-polycytidylic acid (poly I:C) and interferon (IFN)γ. SCD-153 also inhibits LPS-induced increase in secreted IL-1β and IL-6 in murine peritoneal macrophages.
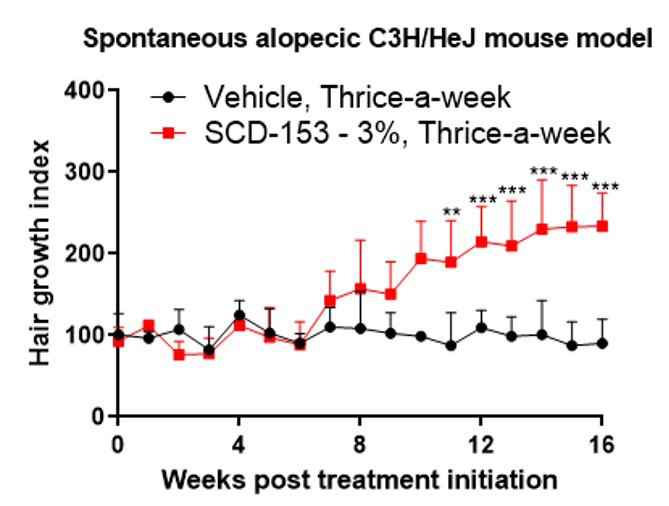
Topical SCD-153 has been shown in vivo to hasten the transition of hair follicles from the telogen to anagen phase leading to hair growth in C57BL/6 mice. Topically applied SCD-153 also induces hair growth in alopecic C3H/HeJ mice, an immune model of human AA, and this hair growth in the treated animals is accompanied by a reduction in pathogenic cytotoxic CD8+ T cells (Tc cells) at the base of the immune-compromised hair follicle.
SCD 153 is currently recruiting patients under Phase I study.
Publication
Topical SCD-153, a 4-methyl itaconate prodrug, for the treatment of alopecia areata. (Tsai J et al) PNAS Nexus; 2023 Jan; 2(1):297.